What Is Alkalic Cleaning For The Epi Polished Wafers
Abstract
A novel arroyo of chemical mechanical polishing (CMP) is developed for cadmium zinc telluride (CdZnTe or CZT) wafers. The approach uses surroundings-friendly slurry that consists of mainly silica, hydrogen peroxide and citric acrid. This is unlike from the previously reported slurries that are normally composed of strong acid, alkali and bromine methanol and are detrimental to the environment and operators. Surface roughness 0.five nm and 4.seven nm are achieved for Ra and tiptop-to-valley (PV) values respectively in a measurement expanse of 70 × 50 μmii, using the developed novel approach. Fundamental polishing mechanisms are also investigated in terms of X-ray photoelectron spectroscopy (XPS) and electrochemical measurements. Hydrogen peroxide dominates the passivating procedure during the CMP of CZT wafers, indicating by the everyman passivation current density among silica, citric acid and hydrogen peroxide solution. Chemical reaction equations are proposed during CMP according to the XPS and electrochemical measurements.
Introduction
Cadmium zinc telluride (CdZnTe or CZT) is a representative for the third-generation soft-brittle semiconductors in room temperature radiations detection, also as a substrate for epitaxial growth of lattice-matched mercury cadmium telluride (HgCdTe or MCT) films used for infrared detectors1,ii,3. Furthermore, CZT is widely used in medical imaging, homeland security and spaceborne X-ray and gamma-ray astronomy1,4,5. This is attributed to its loftier gamma-ray absorption coefficient and high electrical resistivity, derived from the high diminutive number and wide bandgap, respectively1. Nevertheless, CZT has soft and brittle nature6, which is different from the showtime and 2nd-generation semiconductors with hard and brittle characteristics, such as silicon (Si)vii and gallium arsenide (GaAs) respectively, making it a hard-to-automobile material. For instance, the hardness and fracture toughness of CZT are 1.21 GPaeight and 0.158 MPa.thousand0.5 respectively9, which are ane 12th (14.5 GPa)x and ane 6th (0.ix–1.i MPa m0.5)11 those of an Si crystal, correspondingly12.
Surface roughness has a significant effect on the electric property and operation of CZT detectors and therefore atomically smoothen and defect-costless surfaces are necessary to the high-performance CZT-based detectorsxiii,14. Thus, the surface roughness root-hateful-square (rms) <1 nm is required for a high performance CZT detector14,15. For this reason, surface processing techniques for CZT wafers have attracted attentions and are investigated intensively14,15,sixteen,17,18,19,twenty,21. Currently, lapping, mechanical polishing, chemic mechanical polishing (CMP) and chemical etching are usually employed to machine a CZT wafer. Firstly, free abrasives of alumina are widely used to lap and smooth the surfaces of CZT wafers with different grain sizes in a sequence16,17. Nitric acid (HNO3) and bromine methanol (BM) are unremarkably used in CMP14,xviii and chemical etching16,17 respectively, to car CZT wafers. Nonetheless, gratis abrasives of alumina are piece of cake to embed in a CZT surface during lapping and polishing18. Afterwards embedding, the abrasives are hard to remove in the successive processes. This results in the high surface roughness and low surface quality. Moreover, HNO3 is highly corrosive and BM is toxic to the environment and operators. Hereby, information technology is necessary to develop a novel surround-friendly approach of CMP for CZT wafers to overcome the disadvantages of the conventional approaches.
Surface roughness is generally related to the measured area, i.e., smaller area leads to lower surface roughness. For example, surface roughness arithmetic average Ra and PV values of 0.32 and iii.29 nm are reported, respectively using atomic force microscopy (AFM) with a scanning expanse of 0.v × 0.v μm2 on a CZT surfacexiv. With an increase in the scanning area to 5 × five μm2, Ra and PV values increment to 0.94 and 20.2 nm, correspondinglyfourteen. These CZT wafers are polished by HNO3 and BM14. Surface roughness rms of 0.74 nm is obtained on a CZT surface using AFM with a scanning expanse of i × ane μm2 after CMP and chemical etching15. The rms value increases to i.181 nm in a scanned surface of 20 × 20 μm2 which is mechanically polished by free abrasives of alumina, followed by chemical polishing of bromine, ethylene glycol and sodium hydroxide (NaOH)19. Bromine and ethylene glycol are toxic and NaOH is much corrosive. Surface roughness Ra reaches to 1.viii nm measured by AFM with a scanning area of 2 × 2 μm2, which is produced after mechanical polishing by gratuitous abrasives of alumina and diamond, followed past chemical carving of BMtwenty. For decreased toxicity, iodine is dissolved in methanol replacing bromine in chemical carving of CZT wafers subsequently mechanical polishing by diamond pastes and Ra and PV values are 1.563 and 15.85 nm21, respectively with a measurement surface area of 180 × 130 μm2. Notwithstanding, both iodine and methanol are toxic, despite less toxicity of iodine than that of bromine. Surface roughness rms and PV values are ii.063 and 27.834 nm, respectively on the CZT surfaces later mechanical polishing by alumina abrasives16. Surface roughness increases to 3.855 and 95.762 nm, correspondingly after chemic etching using BM. As a result, BM deteriorates the surface roughness, rather than improving it. Undoubtedly, CMP and chemical etching play an of import role to decreasing the surface roughness of the CZT wafers. It is a challenge to develop a novel environment-friendly arroyo to achieve the surface roughness <1 nm with a measurement surface area of 50 × 50 μmii.
In this study, a novel and however surroundings-friendly CMP approach is developed for CZT wafers. The approach uses fixed abrasives of silicon carbide (SiC) in mechanical lapping, followed by CMP consisting of mainly silica (SiO2), hydrogen peroxide (HtwoOtwo) and citric acid. Finally, the polished surfaces of the CZT wafers are cleaned and dried using deionized water and compressed air, respectively. The approach demonstrates promising polishing results of surface roughness Ra and rms values <i nm over a measurement area of 70 × fifty μm2.
Figure 1(a) shows the rough surface of the as-received CZT wafer later on multi-wire sawing. Figure i(b) shows an optical paradigm of lapped surface on a CZT wafer. At that place were neither embedded grains nor cracks, except for micro-scratches. Effigy one(c) shows the polished surface after CMP using the proposed approach. The polished surface looked like a mirror and was perfectly smooth, free of scratches and cracks. The surface roughness Ra, rms and PV values were 0.498, 0.626, iv.707 nm, respectively, with a measurement area of 70.vi × 53 μmii. Therefore, the object of surface roughness rms <one nm is achieved using a novel approach with a measurement area of fifty × fifty μm2.
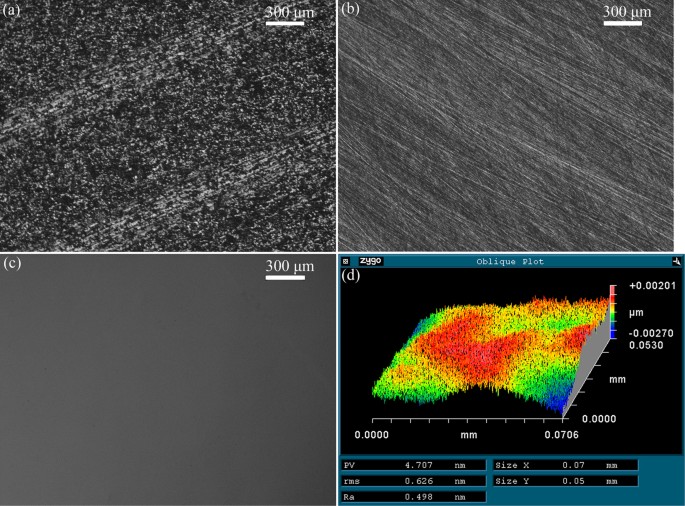
Optical images of (a) as-received, (b) lapped using SiC waterproof papers with mesh size of #2500, (c) polished by the developed approach of CMP CZT wafers and (d) surface roughness and morphology measured by a surface profilometer (NewView 5022) afterward CMP.
Figure two shows the XPS spectra of tellurium (Te) chemical element on different CZT surfaces. All the three surfaces showroom the Te03d5/two, Te4+3d5/2, Te03dthree/2 and Te4+3d3/2 peaks on the as-received CZT wafers, citric acid and mixed slurry consisting of HiiO2, SiO2 and citric acid, as shown in Fig. 3(a,c,d), respectively, except for Teiv+3d5/2, Te4+3d3/2 and extremely weak Te03dfive/2 peaks on the CZT surface induced by H2O2, as seen in Fig. 3(b)22,23.
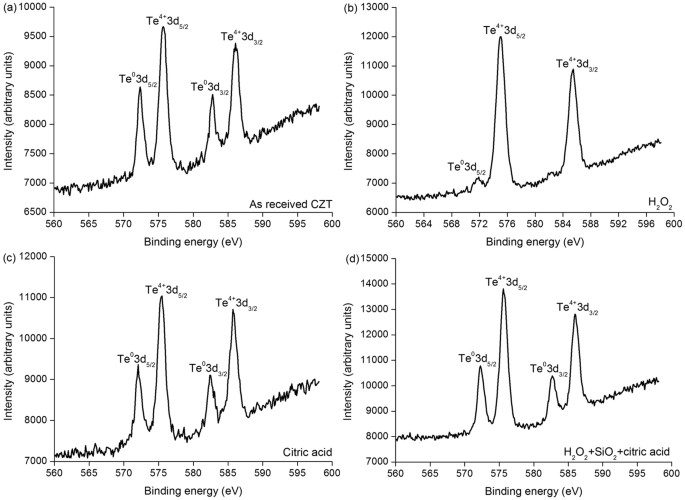
XPS spectra of Te element on CZT surfaces of (a) as-received, (b) HtwoO2, (c) citric acid and (d) mixed slurry consisting of H2O2, SiO2 and citric acid.
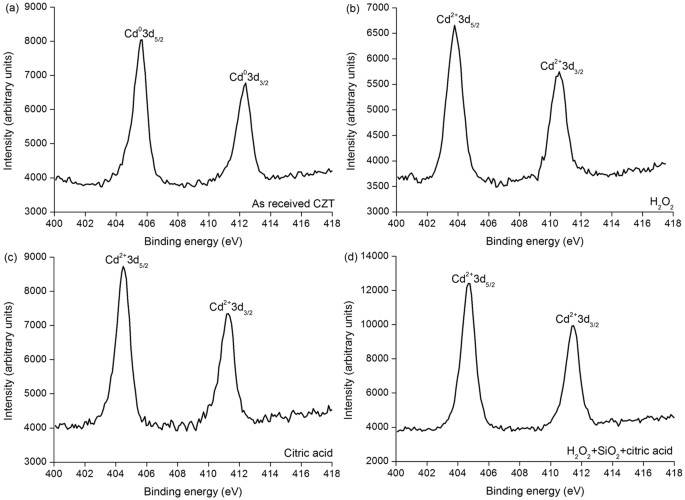
XPS spectra of Cd element on CZT surfaces of (a) every bit-received, (b) HtwoO2, (c) citric acid and (d) mixed slurry including H2O2, SiOii and citric acid.
Figure iii shows XPS spectra of the cadmium (Cd) element on various CZT wafers. The XPS spectra of the every bit-received CZT surface reveal Cd03d5/2 and Cd03d3/2 peaks, as illustrated in Fig. iii(a), which are different from the Cd2+3d5/2 and Cd2+3d3/ii peaks generated by H2O2, citric acrid and mixed slurry of H2O2, SiO2 and citric acid, equally observed in Fig. iii(b,c,d), correspondingly19,24.
Figure 4 shows the electrochemical curves of HiiO2, SiO2, citric acid and mixed slurry fabricated of H2O2, SiO2 and citric acid every bit a role of potential versus SCE. The corrosion potential is also referred to open circuit voltage (OCV). The corrosion potentials of SiOtwo, citric acrid, mixed slurry and H2O2 are −0.31, −0.21, −0.2, +0.05 V, respectively and their passivation current densities are 10−half dozen.5, x−6.53, x−6.77 and 10−6.96 A cm−2, correspondingly, at the potential versus SCE of +0.iv V. At potential versus SCE of +one.2 V, the passivation electric current densities are 10−6.45, 10−6.43, 10−6.17 and 10−6.11 A cm−2, for H2O2, mixed slurry, citric acid and SiOii, respectively25,26.
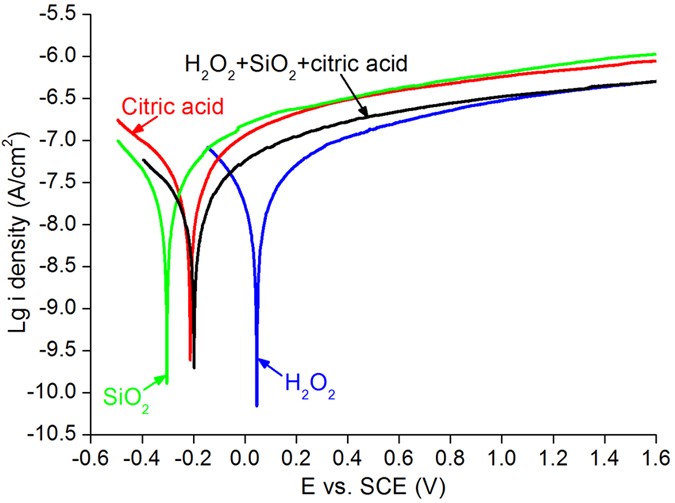
Electrochemical curves of HtwoOii, SiO2, citric acid and mixed slurry made of H2O2, SiOii and citric acid every bit a function of potential versus SCE.
Fixed abrasives of SiC grains are used during lapping processes, which is effective in eliminating the embedding of free abrasives, as shown in Fig. 2(b). The fixed abrasive lapping is different from the previous findings, in which free abrasives are commonly employed. Ultrafine SiC grains, such equally mesh sizes of 2500, 5000 and 8000, are efficient in decreasing surface roughness, saving fourth dimension and price of CMP.
In the CMP slurry, HiiO2 slowly decomposes into water and oxygen gas in air,

H2O2 solution is used as a medical disinfector. In this work, H2O2 solution is diluted by silica slurry and citric acid solution and information technology is environment-friendly. Even the mixed slurry composed of H2O2, SiO2 and citric acid flushes hands and the hands will turn into calorie-free xanthous. After h2o flushing, the calorie-free xanthous color fades. Silica slurry contains SiOii and deionized h2o. SiO2 distributes widely in nature and occupies a weight percentage of 12% in the earth chaff, such every bit stones mainly consisting of SiOii and calcium carbonate (CaCO3). The size of SiO2 spheres played a pregnant consequence on the material removal rate27 and therefore the diameters of SiO2 spheres varied from 25 to 118 nm, as shown in Fig. 5. Mostly speaking, the distribution of particle size is important to controlling surface roughness, i.e. either a high cloth removal charge per unit with big particles, or low surface roughness with relatively small particles. An advisable distribution of particle size could obtain a residual between the material removal rate and surface roughness, resulting in a relatively loftier textile removal rate and low surface roughness. Thus, the distribution of particle size in this study expects to produce coherent outcome for the material removal rate and surface roughness and reach ultralow surface roughness at a relatively high cloth removal rate. Citric acid is a drink and is popular in the nutrient industry. During the terminal stride of cleaning the CZT wafers, deionized water and compressed air are used to displacing the previously toxic etchants and cleaning agents, such as BMtwenty, bromine-based etchants14, methanol19 and ethanol18. Both deionized water and compressed air are natural. Thus, the novel CMP approach for CZT wafers are environment-friendly and tin be applied to lapping, CMP and cleaning processes.

TEM image of silica spheres with diameters ranging from 25 to 118 nm.
Central polishing mechanisms are investigated using XPS and electrochemical measurements. In Fig. 4, the passivation current density of the HiiO2 solution is the lowest among the iv solutions, implying that the nigh compact passivation films tin be formed on the CZT wafers. Citric acid is a pH adjustor in the CMP slurry and its passivation upshot is similar to that of the silica slurry. This is attributed to the similar curves of passivation current density between the citric acid and the silica slurry. In the mixed slurry, the corrosion potential decreases from +0.05 of H2O2 solution to −0.2 V, diluting by SiO2 slurry and citric acid. This leads to decreasing of the pH value of the H2Otwo solution from 2.89 to 7.61 of the mixed slurry, whereas the passivation current density of the mixed slurry is the same as that of the HtwoOii solution after potential versus SCE at +one.2 V. Consequently, H2O2 solution dominates the passivation electric current density, playing a key role in dissolving the CZT wafers and forming an ultrasmooth surface with the lowest surface roughness. Thereby, the chemic reaction equations of the HtwoO2 solution are proposed with CZT wafers28,29,30:


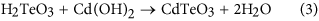


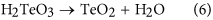
In Figs 2(a) and three(a), Cd03d, Te03d and Te4+3d are present. Cd03d and Te03d are derived from Cd0.96Zn0.04Te surface. Te4+3d valence state comes from the following chemical reaction:
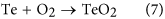
This is because of the as-received CZT wafers are exposed in air afterwards multi-wire sawing from an ingot of CZT. Te usually enriches on the surfaces of the CZT wafers and therefore Eq. (7) prevails. Appropriately, Cd is at the land of Cd03d. With Eqs (2, three, 4, five, vi), the final reaction products are CdTeO3, ZnTeO3 and TeOii nether the function of the HiiO2 solution. This is verified past the Te4+3d valence country in Fig. ii(b). As Cd(OH)2 and Zn(OH)2 are dissolved in H2Oii and mixed slurry with ion state, Eqs (three) and (4) have priority than Eq. (five) for an unproblematic substance. In the high concentration of the H2Otwo solution, Eq. (5) reacts effectively, leading to an extremely small corporeality of Te left, equally illustrated in Fig. two(b). Nonetheless, in the mixed slurry, the H2Otwo solution is greatly diluted to a pH value of seven.61, Eq. (5) reacts ineffectively, resulting in a large amount Te appeared, as drawn in Fig. ii(d). With the effect of HtwoO2, Cd(OH)two and CdTeO3 are produced, which are presented in Eqs (2) and (iii), respectively. This is confirmed past Fig. 3(b,d) in the H2O2 and mixed slurry, correspondingly. Citric acid ionizes hydrogen (H) ions. The following equation happens:
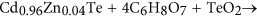
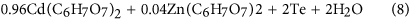
With Eq. (8), Cd2+3d is establish in Fig. 3(c). Citric acid is a pH adjustor, passivation effect is similar to that of the silica slurry, every bit shown in Fig. 4. Equally a consequence, the dissolving effect of the citric acid for the CZT wafers is comparatively weak, resulting in both Teiv+3d and Te03d in Fig. ii(c). On the other mitt, CZT crystals are easy to slip under stress even induced past high speed grinding, rather than forming amorphous phase, in terms of their low stacking fault energy (9.7 ± 1.7 mJ m−2)6,9. In consequence, the polished surfaces of CZT wafers are unremarkably crystalline.
In summary, fixed abrasives of SiC are used in lapping CZT wafers to eliminate effectively the embedded free abrasives to save time and price for subsequent CMP processes. A novel CMP approach is proposed in which the newly developed slurries consist of mainly H2Otwo, SiOii and citric acrid. The novel arroyo is environment-friendly. H2O2 solution dominates the passivation procedure, which is confirmed by electrochemical measurement. With the best passivation effect of HtwoO2 solution amid four solutions, relatively strong Te4+3d peaks and an extremely small Te03d5/2 peak are found.
Methods
The every bit-received Cd0.96Zn0.04Te (111) wafers were x mm in length, 10 mm in width and one.5 mm in thickness, which were grown by the modified Bridgman method1. A precision polisher (YJ-Y380 of Shenyang Yanjia Co., Ltd. China) was employed to lap and shine the CZT wafers. SiC waterproof papers were put on a stainless steel plate as lapping pads. 4 CZT wafers were fixed using a 502 glue on an aluminum plate of 150 mm in diameter uniformly along its periphery. CZT wafers were lapped using SiC papers with mesh sizes in a sequence of 2500, 5000 and 8000 and lapping time was fix at iii, 2, two min, respectively. During lapping, the pressure of the lapping plate was twenty kPa and rotation speeds of both the CZT wafers and SiC papers were 65 rpm. After lapping, the CZT wafers were cleaned using deionized wafer and stale by compressed air for farther characterization by an optical microscope (Olympus).
After the optical label, the SiC papers were replaced by floss polishing pads on the stainless steel plate. The morphology and size of SiO2 spheres were measured by manual electron microscopy (TEM, Tecnai spirit, FEI, Netherlands). The SiOii spheres were used to produce silica slurry with a pH value ranging from 7 to 7.v and a weight percent of 60%. The oxidant was H2O2 solution that had a volume percentage of 40%. A volume ratio of 7 to four between the silica slurry and HtwoO2 solution was used to set the CMP slurry for the CZT wafers. Citric acrid was used as a pH adjustor. The pH value of the CMP slurry varied from 4 to 4.v, which was decreased by the citric acrid. During CMP, the rotation speeds of both the CZT wafers and floss polishing pads were 65 rpm. The polishing pressure and time were 30 kPa and 25 min, respectively. After CMP, the CZT wafers were cleaned and dried using deionized h2o and so compressed air.
Except the optical label, surface roughness and morphology of the CZT wafers were besides measured by a precision non-contact surface profilometer (NewView 5022, Zygo, U.s.). X-ray photoelectron spectroscopy (XPS) was obtained by a VG ESCALAB MKII spectrometer with a magnesium Kα excitation source. Electrochemical measurement was performed on an advanced electrochemical system (PARSTAT 2273, Princeton Applied Research, Ametek, Inc.). The referenced and auxiliary electrodes were saturated calomel electrode (SCE) of potassium chloride (KCl) and platinum (Pt) with purity of 99.99%, respectively. In electrochemical measurement, the pH values of H2O2, citric acid and mixed slurry consisting of H2O2, Sotwo and citric acid were ii.89, 4.45, vii.61, respectively.
Additional Information
How to cite this article: Zhang, Z. et al. A novel approach of chemical mechanical polishing for cadmium zinc telluride wafers. Sci. Rep. half-dozen, 26891; doi: 10.1038/srep26891 (2016).
References
-
Cohen, T. G., Sinkevich, O., Levinshtein, Yard., Ruzin, A. & Goldfarb, I. Atomic structure and electric backdrop of In(Te) nanocontacts on CdZnTe (110) past scanning probe microscopy. Adv. Funct. Mater. xx, 215–223 (2010).
-
Yang, G. et al. Low-temperature spatially resolved micro-photoluminescence mapping in CdZnTe unmarried crystals. Appl. Phys. Lett. 98, 261901 (2011).
-
Ruzin, A., Sinkevich, O., Cohen, T. 1000. & Goldfarb, I. Anomalous beliefs of epitaxial indium nano-contacts on cadmium-zinc-telluride. Appl. Phys. Lett. 101, 132108 (2012).
-
Androulakis, J. et al. Dimensional reduction: a blueprint tool for new radiation detection materials. Adv. Mater. 23, 4163–4167 (2011).
-
Zhang, Z. Y. et al. Deformation twinning evolution from a single crystal in a face-centered-cubic ternary alloy. Sci. Rep. v, 11290 (2015).
-
Zhang, Z. Y., Xu, C. G., Zhang X. Z. & Guo, D. M. Mechanical characteristics of nanocrystalline layers containing nanotwins induced by nanogrinding of soft-brittle CdZnTe single crystals. Scripta Mater. 67, 392–395 (2012).
-
Zhang, Z. Y., Huo, F. W., Wu, Y. Q. & Huang, H. Grinding of silicon wafers using an ultrafine diamond wheel of a hybrid bond material. Int. J. Mach. Tools. Manuf. 51, 18–24 (2011).
-
Zhang, Z. Y. et al. Chemical mechanical polishing and nanomechanics of semiconductor CdZnTe single crystals. Semicond. Sci. Technol. 23, 105023 (2008).
-
Zhang, Z. Y., Zhang, X. Z., Xu, C. Thousand. & Guo, D. M. Label of nanoscale chips and a novel model for face nanogrinding on soft-brittle HgCdTe films. Tribol. Lett. 49, 203–215 (2013).
-
Zhang, Z. Y., Wang, B., Kang, R. Thou., Zhang, B. & Guo, D. M. Changes in surface layer of silicon wafers from diamond scratching. CIRP Ann Manuf Technol 64, 349–352 (2015).
-
Qiao, Y. & Chen, J. Resistance of through-thickness grain boundaries to cleavage bully in silicon thin films. Scripta Mater. 59, 251–254 (2008).
-
Chandra, A. et al. Role of surfaces and interfaces in solar prison cell manufacturing. CIRP Ann Manuf Technol 63, 797–819 (2014).
-
Tari, S. et al. Structural and electronic properties of gold contacts on CdZnTe with dissimilar surface finishes for radiation detector applications. J. Electron. Mater. 43, 2978–2983 (2014).
-
Hossain, A. et al. Novel approach to surface processing for improving the efficiency of CdZnTe detectors. J. Electron. Mater. 43, 2771–2777 (2014).
-
Pelenc, D. et al. Evolution of a method for chemical-mechanical preparation of the surface of CdZnTe substrates for HgCdTe-based infrared focal-plane arrays. J. Electron. Mater. 43, 3004–3011 (2014).
-
Zazvorka, J. et al. Contactless resistivity and photoconductivity correlation to surface preparation of CdZnTe. Appl. Surf. Sci. 315, 144–148 (2014).
-
Duff, M. C. et al. Event of surface preparation technique on the radiations detector operation of CdZnTe. Appl. Surf. Sci. 254, 2889–2892 (2008).
-
Lucile, C. T. et al. Label of etch pit formation via the Everson-etching method on CdZnTe crystal surfaces from the majority to the nanoscale. Nucl. Instrum. Methods Phys. Res. Sect. A 652, 178–182 (2011).
-
Aqariden, F. et al. Influence of surface polishing on the structural and electronic properties of CdZnTe surfaces. J. Electron. Mater. 41, 2893–2898 (2012).
-
Zheng, Q. et al. Influence of surface grooming on CdZnTe nuclear radiation detectors, Appl. Surf. Sci. 257, 8742–8746 (2011).
-
Ivanits'ka, 5. G. et al. Chemical polishing of CdTe and CdZnTe in iodine-methanol etching solutions. J. Electron. Mater. 40, 1802–1808 (2011).
-
Cheng, 10. et al. Effect of surface preparation on the backdrop of Au/p-Cd1−xZnxTe. Appl. Surf. Sci. 253, 8404–8407 (2007).
-
Li, Q. & Jie, W. Q. Surface passivation and electrical properties of p-CdZnTe crystal. Semicond. Sci. Technol. 21, 72–75 (2006).
-
Okwechime, I. O. et al. Chemical handling of CdZnTe radiation detectors using hydrogen bromide and ammonium-based solutions. Proc. SPIE Int. Soc. Opt. Eng. 9213, 92130Y (2014).
-
Feng, Y. Y. & Gu, M. The electrochemical behavior of tellurium on GCE in sol and solutions. Electrochim. Acta 90, 416–420 (2013).
-
Chaure, N. B., Chaure, S. & Pandey, R. Thousand. Cd1−xZnxTe sparse films formed by non-aqueous electrochemical route. Electrochim. Acta 54, 296–304 (2008).
-
Zhang, Z. F., Liu, W. Fifty. & Vocal, Z. T. Particle size and surfactant effects on chemical mechanical polishing of drinking glass using silica-based slurry. Appl. Opt. 49, 5480–5485 (2010).
-
Dean, J. A. Lange's handbook of chemistry 15th ed. New York, McGraw-Colina Company 8.124–8.139 (1999).
-
Wang, 10. Q., Jie, W. Q., Li, Q. & Gu, Z. Surface passivation of CdZnTe wafers. Mater. Sci. Semicond. Process. 8, 615–621 (2005).
-
Chen, S. C. Important inorganic chemical reactions tertiary ed. Shanghai, Shanghai Printing of Science and Technology p. 947 (1994).
Acknowledgements
The authors are grateful for the contribution of CMP from Master Yaxing Song graduated from DUT. Z.Y.Z. thanks to the valuable discussions with Prof. Kang Shi at Xiamen University. The authors are grateful for the fiscal support from the Excellent Young Scientists Fund of NSFC (51422502), Integrated Program for Major Inquiry Program of NSFC (91323302), Science Fund for Creative Research Groups of NSFC (51321004), Program for New Century Excellent Talents in University (NCET-thirteen-0086), the Fundamental Research Funds for the Central Universities (DUT14YQ215), the Tribology Science Fund of State Key Laboratory of Tribology (SKLTKF14A03), Tsinghua University, the Science Fund of the State Fundamental Laboratory of Metastable Materials Science and Technology (201501), Yanshan University, the Xinghai Science Fund for Distinguished Young Scholars at Dalian University of Technology, the Outstanding Cosmos Talents "Cloud Projection" of Changzhou City (CQ20140008) and the Natural Science Foundation of Jiangsu Province (BK20151190).
Author information
Affiliations
Contributions
Z.Y.Z. and D.M.G. conceived the projects. Z.Y.Z. designed the experiments. Z.Y.Z. and B.W. carried out the experiments. Z.Y.Z. and B.Z. co-wrote the paper. Z.Y.Z., B.W., P.Z. and R.K.K. analyzed the fundamental mechanism of chemical mechanical polishing for CZT wafers. All authors discussed the results and commented on the manuscript.
Ethics declarations
Competing interests
The authors declare no competing fiscal interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution iv.0 International License. The images or other 3rd party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the cloth is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/past/4.0/
Reprints and Permissions
About this article
Cite this article
Zhang, Z., Wang, B., Zhou, P. et al. A novel approach of chemical mechanical polishing for cadmium zinc telluride wafers. Sci Rep 6, 26891 (2016). https://doi.org/10.1038/srep26891
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/10.1038/srep26891
Farther reading
Comments
Past submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it every bit inappropriate.
Source: https://www.nature.com/articles/srep26891?error=cookies_not_supported&code=318d15fc-f5a1-4abf-8988-d0114736b2d7
Posted by: dejongbuther.blogspot.com


0 Response to "What Is Alkalic Cleaning For The Epi Polished Wafers"
Post a Comment